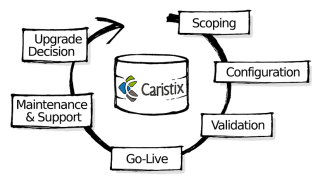
 Last week, we talked about how you can do more with less by applying a lifecycle approach to your interface and interoperability work. By managing the lifecycle (instead of developing via trial and error), you’re able to forecast timelines, resources, and costs with more accuracy – and better outcomes and margins.
Last week, we talked about how you can do more with less by applying a lifecycle approach to your interface and interoperability work. By managing the lifecycle (instead of developing via trial and error), you’re able to forecast timelines, resources, and costs with more accuracy – and better outcomes and margins.
Start by getting a firm grasp on the key interface lifecycle concepts behind interface lifecycle management. This post will give you a solid foundation.
HL7 requirements
You need requirements thinking to build an interface. In other words, you document the needs that the interface must meet: messages, content, format, workflows. Sounds simple, right? Not really. Traditional requirements-gathering leads to trial and error pain, slowing down both system and interoperability implementations.
Learn about the right way to gather HL7 requirements.
Reverse-engineering
HL7 requirements include accurate specifications of the systems you plan to connect. What do you do if the vendor spec is missing? Or if a PDF spec no longer matches the production version? There’s a way to generate the spec automatically from production messages, so you get a list of system-specific attributes such as message types, segments, fields, data types, and code sets. It’s called reverse-engineering, and it’s a technique that we developed at Caristix.
Learn about reverse-engineering and how it can impact your interface development.
Gap analysis
Another requirement document you need for building an interface is a list of gaps the interface will bridge: a gap analysis. A good gap analysis will map differences in message structure and content in the source and destination systems. You can either do this work manually or use automated software to speed the process and gain accuracy.
De-identification
De-identification is a technique to mask or remove PHI from patient data. Surprisingly, it’s a life-saver in HL7 interfacing. You’ll want to consider de-identifying HL7 messages whenever you’re faced with troubleshooting a system or interface, populating a test system with realistic messages, and gathering data for analytics.
Learn about de-identification.
Validation
Validation is about checking the interface: does it meet the requirements you captured at the start of the project? Traditionally, validation is done manually: creating test messages, sending them, and checking the workflow. But manual work can be a recipe for disaster, which is why we recommend automating validation and testing tasks.
Learn more about validation and automation.
Other concepts?
Can you think of other concepts we should cover in an upcoming blog post? Leave a comment to let us know.
For a review of the key phases of interface lifecycle managment, check out our last blog post, Interface lifecycle managment: do more with less.
